
Temprature Mapping Validation Services
Product Details:
X
Product Description
About Pmall-Validation Services:
Few Application of Temperature Mapping in Various Indsturies:
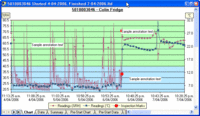
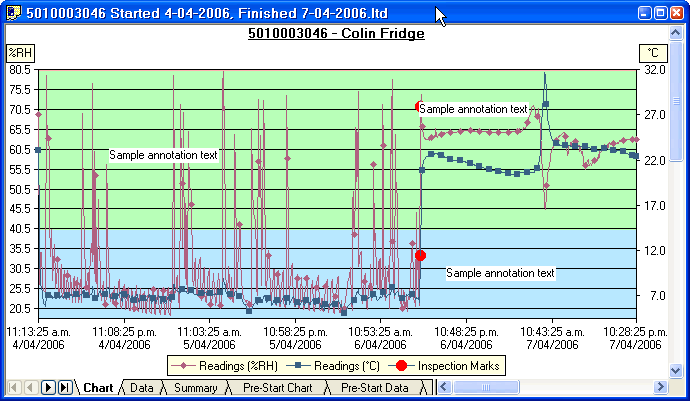
Stability Chambers/BOD/Incubator and Warm Storage:
- Pmall-offers Validation Services that meet WHO, cGMP/ USFDA etc.. standards.
- We Customize our validation protocols to meet the specific quality system of our customers.
- Our customers review and accept our documents before we move a project forward to ensure that there are no gaps in documentation or expectations, and all outcomes are met.
- Our services can be protocol development only, execution of a customers protocols, or turnkey protocol development and execution.
- A validation project manager will assist our customers in defining the scope of work for each project and provide a detailed quote that clearly establishes the scope of the project.
Few Application of Temperature Mapping in Various Indsturies:
- Cold Storage/ Area Temperature Mapping
- Autoclave/ DHS / Steriliser Validation
- Oven Validation
Stability Chambers/BOD/Incubator and Warm Storage:
- Temperature mapping of both empty and full chambers
- Calibration of pressure gauges and temperature probes and time are in each validation
- Loaded, or empty chamber profile mapping are available
- Flexible mapping times as defined by customer requirements
- Use of NIST/NABL traceable Sensors or standard data acquisition systems for all projects
- Our customers review and accept our documents before we move a project forward to ensure that there are no gaps in documentation or expectations, and all outcomes are met.
Enter Buying Requirement Details
Other Products in 'Validation Services' category
 |
PRISM TEST AND MEASURE PRIVATE LIMITED
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |